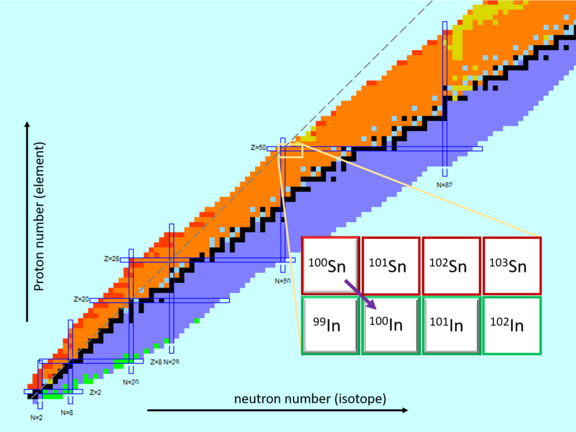
The tin isotope with the mass number 100 is the heaviest possible atomic nucleus with the same number of protons and neutrons. The properties of this atomic nucleus are a "Holy Grail" of nuclear physics. With “magic” number 50, the 50 neutrons and 50 protons fully occupy their respective shells and tin-100 is therefore a doubly magic nucleus. Actually, doubly magic nuclei are particularly stable, but for heavy nuclei like tin-100 this is only true in comparison to their neighbouring nuclei. Tin-100 is short-lived and decays to indium-100 (with 49 protons and 51 neutrons). This extremely exotic nucleus is therefore very difficult to produce in sufficient quantities and largely eludes direct, precise measurements. In fact, there are presently two contradictory values for the decay energy of tin-100 in the literature.
To learn more about the properties of this special nucleus, theoretical calculations provide a possible avenue. But how reliable are the values obtained in this way? This can be checked using precise mass measurements of neighbouring nuclei in the nuclear chart.
At the ISOLDE isotope separator at CERN, it was possible to produce the indium isotopes 99, 100 and 101 (the latter in the ground state and an excited metastable state), which are also quite short-lived and neutron-deficient, separate them and feed them to the ISOLTRAP mass spectrometer. This consists of a time-of-flight instrument and a downstream Penning-trap system, with which the experimenters determined the masses of these nuclei with high precision. From this, they deduced their binding energies – because the mass of an atomic nucleus is the sum of the masses of the contained nucleons, i.e., the protons and neutrons, and the binding energy. The value for indium-100 and the literature values for the decay energy result in strongly contradictory values for the binding energy of tin-100.
In parallel, the theory team performed calculations of indium, tin and neighbouring atomic nuclei around the "Holy Grail" tin-100 using state-of-the-art ab initio methods and two- and three-nucleon interactions. The results of all methods for indium as well as for more neutron-rich tin isotopes show the same trends and agree well with the experimental data. This makes the predictions for tin-100 very trustworthy. Somewhat surprisingly, both the theoretical predictions and the experimental data support the older and not the newer, actually more accurate decay energy measurement.
Original publication:
Mass measurements of 99–101In challenge ab initio nuclear theory of the nuclide 100Sn,
M. Mougeot, D. Atanasov, J. Karthein, R.N. Wolf, P. Ascher, K. Blaum, K. Chrysalidis, G. Hagen, J.D. Holt, W.J. Huang, G.R. Jansen, I. Kulikov, Yu.A. Litvinov, D. Lunney, V. Manea, T. Miyagi, T. Papenbrock, L. Schweikhard, A. Schwenk, T. Steinsberger, S.R. Stroberg, Z.H. Sun, A. Welker, F. Wienholtz, S.G. Wilkins, and K. Zuber,
Nature Physics 23.09.2021, DOI: 10.1038/s41567-021-01326-9